University of Iowa biologists have definitively linked the brain's immune system to epilepsy.
In a new study, the researchers lay out a chain of events that can cause seizures—the most common manifestation of epilepsy—to worsen. The sequence begins when oxidative stress in the body causes the brain’s immune system to react. That activation by the brain’s resident immune cells (called glia) triggers more severe seizures.
The findings are important because it’s the first time the brain immune system–epilepsy link has been experimentally proved. That advance should yield more precise testing and more effective medications to treat the condition.

“We have provided genetic proof that both oxidative stress and activation of the brain immune system make epilepsy worse,” says John Manak, professor in the Department of Biology and Stead Family Department of Pediatrics at Iowa and the study’s corresponding author. “This is hugely significant because our data suggest that we can now repurpose exceedingly well-tolerated anti-inflammatory compounds as well as perhaps antioxidants to help control epilepsy progression.”
Epilepsy affects 3.4 million people—the vast majority being adults—in the United States, according to the U.S. Centers for Disease Control and Prevention. Worldwide, an estimated 5 million people are diagnosed with epilepsy each year, according to the World Health Organization.
The researchers clinched the brain–epilepsy connection in experiments using fruit flies. One reason they chose the insects is because fruit flies and humans share a common ancestral gene, called prickle, that when mutated, leads to seizures. The prickle gene’s involvement in epileptic seizures was determined by, among others, Alex Bassuk, pediatric neurologist and chair of the Stead Family Department of Pediatrics at Iowa, who published findings in 2008 and is a co-author on this study.
Manak’s team also chose fruit flies for their experiments because flies, unlike humans and other vertebrates, have a single, primitive disease response mechanism, called the innate immune system, that is utilized by glial cells in the brain. Vertebrates also have an innate immune system, but that is complemented by an adaptive immune system that involves an army of immune cells with stored “memories” of past, invasive pathogens. The fruit fly’s lone innate immune system meant the researchers could concentrate exclusively on the brain innate immune system–epilepsy connection.
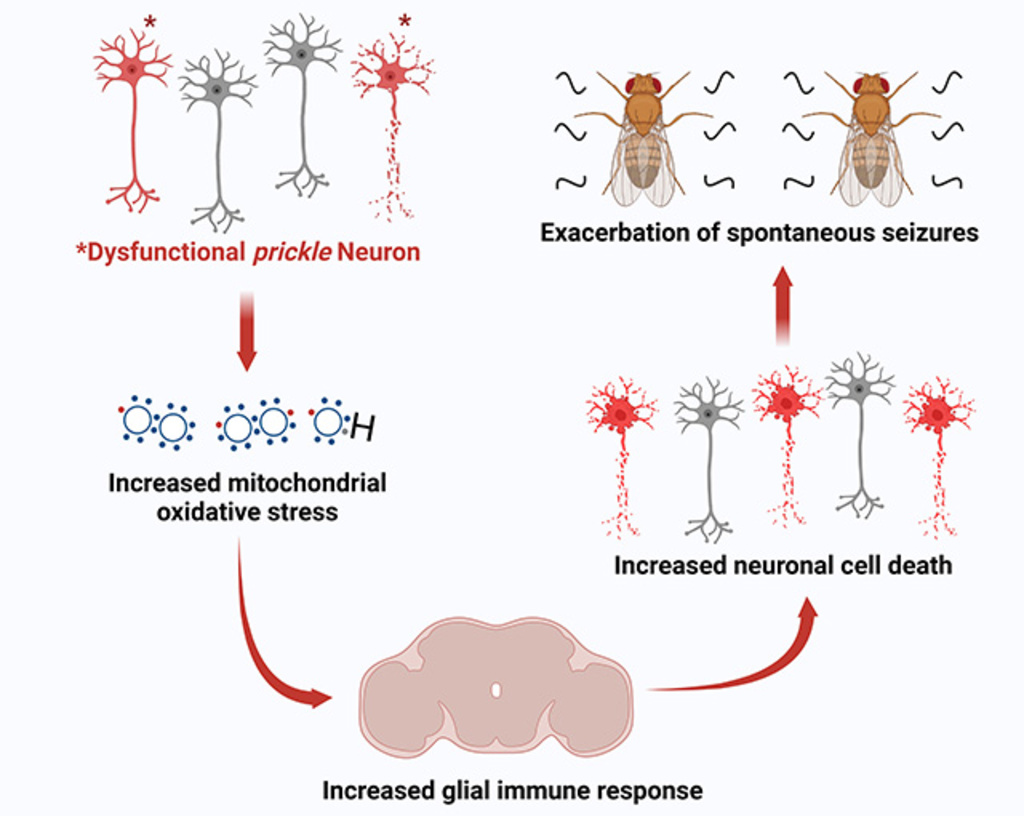
The researchers identified all genes that became expressed in fly brains with seizures (triggered by the mutated prickle gene) compared with those with no seizures. They identified two classes of upregulated genes associated with the seizure-laden flies: immune response genes and those involved in mitigating oxidative stress.
In further experiments with fruit flies, the researchers turned off the innate immune system in the brain glia. That action reduced neuronal cell death, which in turn suppressed seizures.
In another set of experiments with flies, the researchers tested the oxidative stress link by expressing the brain’s SOD1 gene, which is known to cleanse cells of reactive oxygen species caused by oxidative stress. When this gene was activated in the fruit fly brains, the flies’ innate immune system response was lowered, and seizures were reduced.
“In one fell swoop, we have identified a pathway starting at increased oxidative stress leading to activation of the innate immune system, which leads to neuronal cell death, which, in turn, causes exacerbation of seizures,” says Manak, who calls the study one of the highlights of his almost 40-year career as a biologist. “And when I say exacerbation of seizures, I mean progressive epilepsy, which are epilepsies that get worse over time.”
The results mean the flies can be used to directly test which anti-inflammatory drugs or antioxidants would be most effective in treating epileptic seizures. Until now, doctors had only limited evidence that a small number of anti-inflammatory or antioxidant medications had seizure-suppressive characteristics in some contexts, but no direct proof connecting oxidative stress and innate immune system activation with epilepsy. Moreover, approximately one-third of epilepsy patients do not respond well to currently available therapies, and two-thirds of patients have adverse side effects to the medications, according to the study’s authors.
“Not every anti-inflammatory or antioxidant compound is going to effectively treat epilepsy,” Manak explains. “We now have the perfect model with our flies to screen through a significant number of anti-inflammatory and antioxidant compounds. We can then elevate any promising drugs to mouse models, and then potentially human trials.”
The study, “Downregulation of oxidative stress-mediated glial innate immune response suppresses seizures in a fly epilepsy model,” was published Jan. 31 in the journal Cell Reports.
The first author is Krishna Nukala, who earned a doctorate in integrated biology at Iowa in 2022 and is now a senior scientist at Thermo Fisher Scientific. Co-authors include Anthony Lilienthal, a graduate student in biology at Iowa; Shu Hui Lye, from the University of Alabama-Tuscaloosa; Bassuk; and Stanislava Chtarbanova, from the University of Alabama-Tuscaloosa.
The National Institutes of Health funded the research.
